Dairy epicenter of broader FDA strategy
Public comments due August 27
By Sherry Bunting, from Farmshine August 10, 2018
WASHINGTON, D.C. — While the dairy industry is focused on multiple layers to the milk and dairy standards of identity — the FDA review of these standards, and their enforcement, is couched within the broader Comprehensive Multi-Year Nutrition Innovation Strategy launched recently by the Food and Drug Administration (FDA).
Labeling and education are aspects of the strategy, along with a new FDA definition of “healthy choice” to be identified “visually” on foods that meet criteria FDA is still defining.
But the key to the strategy, according to FDA, is to “modernize” standards of identity in order to achieve specific nutrition goals the agency believes will reduce chronic diseases – namely diabetes, obesity and heart disease.
Since 80 of the 280 foods with current standards of identity are dairy foods, and many of the remaining 200 are meat products, the FDA’s noted allegiance to the low-fat dogma of the current Dietary Guidelines does not bode well for how this may all turn out.
That’s why grassroots consumers and producers NEED to get involved.
Of particular concern in a July 26 FDA hearing are regulator use of terms such as “barrier to innovation” or “reformulations of foods using science” in discussing how modernized standards of identity can help the government attain an objective of getting consumers to eat in accordance with the ways it believes will lower chronic disease.
This, despite the fact that noted scientists in the health and nutrition fields and investigative science journalists, like Nina Teicholz, author of The Big Fat Surprise, continue to point out how flawed the science has been for current dietary guidelines, and how these flawed guidelines have actually led to epidemic rates of obesity and diabetes while doing nothing to abate heart disease and morbidity.
Teicholz observed in submitted comments that, “There is no evidence that saturated fats cause obesity. Consumption of saturated fats have declined 17% since 1970, animal fats down by 29% in same period, while obesity rates are up, so explain how saturated fats can be the cause?”
Hearings on parts of the Nutrition Innovation Strategy have already taken place prior to FDA commissioner Scott Gottlieb’s now-famous statement that “Almonds don’t lactate.” This statement propelled milk and dairy into the epicenter of the standard of identity modernization process FDA already had in motion.
A daylong FDA hearing on July 26 kicked off the standard of identity portion of the Nutrition Innovation Strategy, and two weeks prior, the administration held a listening session specifically on the labeling and regulation of new cell-cultured protein technologies — funded by billionaire investors and conventional agriculture companies — seeking to gain standardized status as ‘animal-free’ versions of various meats and dairy proteins for inclusion in products — interchangeably without notice.
The July 26 session attracted a larger than expected attendance due to the national discussion on imitation milk products, and FDA moderator Kari Barrett indicated there was a “very large webcast audience participating.”
Commissioner Gottlieb kicked it off telling how FDA has been monitoring food innovation trends and sees these trends as providing an opportunity to empower individuals to use nutrition to reduce chronic disease.
He acknowledged a “deep personal interest” in the Nutrition Innovation Strategy as he believes nutrition innovation can help solve health issues, and he believes FDA can develop a policy framework to achieve it in their regulatory role.
(However as the daylong hearing progressed, it became obvious that the notable presenters and regulators on various panels are relying heavily on the current flawed Dietary Guidelines for Americans, which themselves are in need of “modernization” due to the revelations about the poor science behind them, particularly on saturated fat).
“We want to empower consumers with innovation and facilitate industry innovation for healthier foods … to remove barriers and leverage nutrition toward these goals,” said Gottlieb, calling it one of his “top priorities.”
He reminded participants that FDA regulates 80% of the food supply with a long history of informing that regulatory process via the Dietary Guidelines for Americans.
“We want to modernize our regulatory approaches to help consumers seek healthier options,” said Gottlieb. “The historic approach (of FDA) is to set barriers. But by modernizing our framework and approach and looking at consumer trends, the food industry can provide these healthier options with foods consumers are seeking.”
The new area of focus for the agency, according to Gottlieb, will be to see the food industry “compete on the nutritional attributes of their products” within a policy framework that allows innovative reformulation.
Gottlieb also mentioned “calls for FDA to take a closer look at dairy identity,” he said. “But first we must better understand how consumers understand and use the term milk and how they are being misled.”
Gottlieb acknowledged the “proliferation of beverages calling themselves milk” and said the FDA is being questioned about its enforcement of milk’s standard of identity.
“The challenge is that we can’t unilaterally change if we have been historically enforcing it a certain way,” he said. “That’s what we are starting, a conversation. We are meeting with interested stakeholders and will post a definition later this summer or early fall with specific questions for feedback and then revisit our enforcement.”
The next steps after comments, feedback and proposed definitions for milk will be to provide the industry with guidance on labeling, and then compliance.
“In the meantime, we will take steps on labels where there is a high likelihood of consumers being misled in cases where public health is affected,” Gottlieb said.
In total, the FDA has 280 standards of identity on the books “created when our grandparents were younger than me,” he said. “We want to hear about the changes in science to review so that we can update these standards.”
He gave the example of standards of identity being modernized with industry and consumer input “to reduce fat and calories.”
He said that FDA wants “to gather input and encourage out-of-the-box thinking” so that the standards are not so rigid as to “cause the industry to avoid reformulations that would reduce fat and calories.
“We need diverse opinions,” said Gottlieb, “but the bottom line (of the Nutrition Innovation Strategy) is for consumers to identify healthier options.”
He said that, “Disparities in diet contribute to disparities in health… modernizing standards and label claims are a key element of our strategy to give consumers quick signals with important information on nutritional benefits and provide incentives for industry to innovate for foods with more healthful attributes.”
Expert panelists, like David Portalatin, vice president and food industry advisor, The NPD Group, testified about consumer trends: “Our data suggest that plant-based protein alternatives are increasing very rapidly, and a large percentage of these consumers are not vegan.”
“Protein is the number one thing consumers seem to want to add to their diets, and we’ve seen a proliferation of ways to add it, and consumers say ‘yes’ I’ll try that,’” said Portalatin about the renewed interest in high protein diets. “When we invest in new stuff, we buy it, we are not a meat-avoidance society.”
He noted that according to survey data, 84% of people reporting they are consuming plant-based alternatives while they are not vegan or vegetarian. “There are a lot of us trying these alternatives,” said Portalatin.
He also mentioned that the interest consumers have in purchasing “low-fat” foods is declining, that people want real food – as it is – and want to control their intake of fat by portion size.
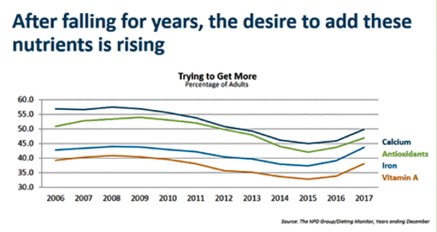
David Portalatin said that in addition to more protein and fewer concerns among consumers about fat, consumers aso want more calcium, iron, Vit. A and antioxidants. Milk contains all and is a big source all but iron, while beef is a big source of the iron. A recent study showed that milk and other dairy foods are “densely packed” with antioxidants delivered in a more soluble way via the protein and fat found naturally in milk — the very fat that FDA and the food police want people to eat less of and the very protein that some experts at the hearing said consumers want more of, but are not deficient in. In other words, consumers are going one way with their diets (away from flawed guidelines that have led to chronic illness) while the government may use this nutrition strategy to shepherd consumers back into the flawed guidelines obedience flock they are just now breaking free of. Screenshot by Sherry Bunting during FDA hearing webcast
Interestingly, the four areas consumers are trying to improve their diets are found in the combination of real milk and beef in the diet.
The American Heart Association had a representative during the open comment time telling FDA they “want the standards of identity to be modernized to improve the nutritional value of food by reducing sodium and saturated fat.” (More on this in a future part of this series).
While he noted that generational cohorts are the biggest drivers of change, the renewed interest in high protein diets is, in his opinion, not necessary since “American diets are certainly not deficient in protein,” in Portalatin’s opinion.
He and other panelists tended to lump this high protein diet preference with a return to higher fat in the diet. Not one panelist recognized the revelations about low-fat dogma of 40 years contributing to the very chronic diseases the FDA strategy seeks to prevent. Every indication from this hearing is that FDA will fall in line with the Dietary Guidelines Advisory Committee in continuing to tweak all kinds of rules and regulations to get Americans to eat less fat — unconsciously — through modernized standards and reformulated foods.
The Good Food Institute — an organization representing plant-based and cell-culture imitation meat and dairy products — had legal representatives testifying on July 26 that, “FDA’s practice for the last decade or more in its guidance for ‘milk’ is that we could use the term ‘milk’ with appropriate modifiers, like almondmilk. The same has been true of butter, such as cashewbutter.”
The Good Food Institute insisted that when their foods can use a standardized term with a modifier, it allows their new – and they say healthier (but are they?) – products come to market more easily. “If that changes, it will make it harder for newer and healthier foods to come to market,” they said.
A representative for Dairy Farmers of America spoke during the open comment time saying that, “The current standard for milk should be enforced as it is. The plethora of products are borrowing the dairy industry’s nutrition profile, and those products may not be as nutritional or wholesome with FDA not enforcing the standard.”
From the Academy of Nutritionists and Dieticians, Jeannie Blankenship, said their professional members will have to “translate” whatever FDA decides on these things. “Consumers must be able to readily understand,” she said. “People with food allergies and intolerances use these standardized terms in a different manner than the general population.”
North Carolina Ag Commissioner Joe Reardon cited the standard of identity defines milk as a lacteal secretion of the mammary gland. “Plant-based beverages do not meet that definition,” he said. “If milk is on the label, then milk should be in the product. Without enforcement of this simple standard, then all standards of identity are compromised.”
He and others made it clear they are not advocating for these plant-based beverages to be removed from the market.
“We recognize they are a vital option for many consumers; however, they should be labeled correctly, without the term ‘milk,’” said Reardon. “North Carolina and other states stand ready and willing to assist FDA to enforce this standard and for the industry to come into compliance. We have heard here all day about the importance of a label, but without truth in labeling, none of the other matters.”

Kim Bremmer testified to FDA on behalf of the American Dairy Coalition about enforcement of the definition of milk. Photo provided by ADC
Speaking for the American Dairy Coalition, which recently started an Integrity Initiative, Kim Bremmer, a dairy producer from Wisconsin said “You play a vital role in giving consumers the information to make choices. Nutrition matters. I see tens of thousands of consumers in my speaking and the vast amount think some of these beverages have cow’s milk in them, and most believe they are as nutritional as milk, and they aren’t. The play on words is misleading.”
Bremmer described cow’s milk as a powerhouse of nutrition with crucial nutrients for cellular function. “No other drink packs this nutrition. There’s no comparison,” she said, explaining what she sees and hears when fourth-graders visit her farm and she watches the children connect the dots to realize the almondmilk they may be drinking at home, isn’t milk at all.
“One in five people are food insecure and one out of 10 adolescent girl are deficient in calcium. We have a problem. We must protect the integrity and identity of milk because nutrition matters,” said Bremmer.
Rob Post from Chobani was there to talk about getting a standard of identity for Greek yogurt so that schools and other institutional feeding situations could accurately quantify the protein levels. As it is now, they are standardized at the regular yogurt levels of protein even though strained Greek yogurts are 52% protein — twice that of regular yogurt.
While he said standards of identity have not kept pace with new food innovations, and he wants to see a better process, he was quick to defend the current definition of milk and dairy — and its enforcement — saying that, “It’s important to have options, but words matter to consumers and dairy means something specific. It means nutrient dense, minimal processing. It is important that this standard is preserved,” said Post.
From National Milk Producers Federation (NMPF), Tom Balmer noted that the issue comes down to “safeguarding the standards to help maintain honesty in the markets.”
“Milk, yogurt, cheese, butter. Standardized dairy terms are being coopted by others as purely a marketing gimmick, while these products lack the nutrients and attributes of dairy,” said Balmer.
“Consumers don’t realize they are being shortchanged. It’s hard to talk about ‘modernizing’ standards when current standards are not enforced. FDA may have forgotten the standard for milk, but we haven’t. Enforce the current standards and stop the confusing and deceitful marketing practices.”
International Dairy Foods Association (IDFA), representing milk and dairy processors, was mum on milk, but touted an array of expanded and modernized standards they want to see for many dairy standards. More on that in part two.
The American Dairy Coalition is urging the FDA to stop allowing the wrongful use of the word “milk” on non-milk, plant-based alternative products labels. To sign the ADC Milk Integrity Initiative petition, it is available online at http://www.americandairycoalitioninc.com/the-integrity-initiative.html
Public comments can be sent to FDA prior to the Aug. 27, 2018 deadline at the docket portal at https://www.regulations.gov/docket?D=FDA-2018-N-2381.
Or, send to: Dockets Management Staff (HFA–305), Food and Drug Administration, 5630 Fishers Lane, Rm. 1061, Rockville, MD 20852
All submissions received must include the Docket No. FDA– 2018–N–2381 for ‘‘FDA’s Comprehensive, Multi-Year Nutrition Innovation Strategy.’’
Look for part 2 this week.
-30-