By Sherry Bunting, updated from original publication in Farmshine, Sept. 30, 2022
WASHINGTON — Get ready for unscientific nutrition bullying. Announced more than a year ago, the White House Conference on Food, Nutrition and Health Wednesday, September 28 was cloaked in secrecy until the eve of the event, when the 44-page “Biden-Harris Administration National Strategy on Hunger, Nutrition, and Health” was released Tuesday, September 27 around Noon.
By 5:00 p.m., the Conference agenda appeared in the inbox of registered participants, and during the overnight hours, the Biden Administration released a fact-sheet announcing $8 billion in “new commitments” from over 100 private businesses, local governments and philanthropies for what it calls a “transformational vision.”
Taking a page from the World Economic Forum’s (WEF) Davos-style approach to food transformation, the White House solicited pledges to address the five “pillars” in its playbook.
Of note among them are a $500 million investment by Sysco (foodservice vendor), nearly $50 million by Danone, $250 million from a collaboration of the Rockefeller Foundation and the American Heart Association on a ‘food as medicine’ initiative, and an undisclosed amount for a collaboration between Environmental Working Group, the James Beard Foundation, the Plant Based Foods Association and the Independent Restaurant Coalition to prompt more plant-based alternative and vegan offerings in foodservice — to name a few.
Then, at 9:15 a.m., just 15 minutes before USDA Secretary Vilsack was set to open the Conference ahead of President Joe Biden’s remarks, the Food and Drug Administration (FDA) announced its “proposed updated definition of a ‘Healthy’ claim on food packages to help improve diet and reduce chronic disease.”
Presto: FDA provided the ‘teeth,’ describing its proposal as aligning directly with the Dietary Guidelines. For the proposed rule, click here and to submit a comment by Dec. 28, 2022, (now updated as comment period ends Feb. 16, 2023): click here
This morsel had been under development over the past four years after public hearings in 2018-19 were reported by Farmshine and then deliberations went silent – until now.
The flurry of activity appeared in scripted fashion within the 24-hours prior to the start of the White House Nutrition Conference convening stakeholders. The first such conference was over 50 years ago and had served as the launch pad for what are known today as the infamous Dietary Guidelines for Americans (DGAs).
A Senate nutrition hearing exactly one year ago in November 2021 paved the way for the September 2022 White House Nutrition Conference.

The Conference and follow up actions, said President Biden on Sept. 28, are being devoted to “nourishing the soul of America so that no child goes to bed hungry and no parent dies of a disease that can be prevented. We can do big things,” he said about the stated 2030 goals of ending hunger, increasing healthy eating and physical activity, and reducing diet-related illnesses and other nutrition-related health inequities.
“But,” Biden declared: “We have to give families a tool to keep them healthy. People need to know what they should be eating, and the FDA is already using its authority around healthy labeling so you know what to eat.”
The President continued: “We can use these advances to do more to be a stronger and healthier nation, to achieve ambitious goals. We must take advantage of these opportunities when we have these children in a whole of government, whole of society approach. We need to think in ways we never thought before.”

In his remarks ahead of the President, Ag Secretary Tom Vilsack stated that government programs feed 1 in 4 children. He and Biden both talked about expanding the child credit permanently. They talked about $2 billion in funding for food banks and schools, including $100 million for ‘incentives’ to make school meals healthier. They both noted funding to make free school meals available for 9 million additional children. A laundry-list of throwing money at a problem without re-evaluating the flawed guidelines that run the school meals and other USDA food programs despite preponderance of evidence that saturated fats are not the enemy.
There was talk of going “a new direction” but this is all process-based. There was no talk of reviewing the flawed Dietary Guidelines that helped get us here and that the Biden-Harris strategy puts so much emphasis on.
Parsing through the 44-page National Strategy, the bottom line is to expect more of the same drill-down on eliminating animal fats, only worse and with stiffer process, labeling and speech boundaries through FDA and the FTC.
We can expect nutrition bullying to commence — if we step outside of the still-vague but Dietary Guidelines-centered White House playbook. In fact, in addition to the FDA ‘Healthy’ label update, a small-print detail in the 44-page Strategy promises power and funding to the Federal Trade Commission (FTC) to scrutinize and penalize food marketing claims for being out-of-bounds on the Biden-Harris DGA-scripted nutrition field of play.
Vilsack noted the National Strategy’s approach is a “whole of government approach that involves the entire federal family.”
In preparation for the Conference, many have lamented the lack of transparency leading up to it. For months, the Conference website gave instructions on how to hold a ‘watch party,’ or a ‘satellite event,’ and how to rally support for nutrition and health ahead of time. But all of the necessary details were missing — until the day of the conference.
Emailed invitations were sent to those who registered just three days before — requesting that they visit a web-portal and record an interview to provide input. There, people respond to White House questions and their faces are added to a streaming screen full of moving mouths — giving the appearance of broad input flowing in from Americans.
Made nervous by the lack of a published agenda or framework, over a dozen agricultural organizations had sent a letter to President Biden on September 8th asking for a “seat at the table.” Those organizations included American Farm Bureau and commodity groups for wheat, beef, sorghum, peanuts, canola, soybeans, barley, corn, sunflower, eggs and rice.
Dairy organizations were conspicuously absent from any of the pre-Conference letter-writing or other such public statements. But then, the dairy industry has its man Vilsack in play, and its DGA 3-a-day – so case-closed – can’t be bothered on the milkfat and whole milk issue.
On the agenda provided the day of the Conference, we found former DMI vice president of sustainability, Erin Fitzgerald — who now serves as CEO of the U.S. Farmers and Ranchers Alliance and who represented USFRA and referenced her boss at the dairy checkoff during a WEF panel in Davos earlier this year — leading a plenary session on “access to affordable foods.” Also, Chuck Conners of the National Association of Farmer Cooperatives led the plenary discussion on “empowering consumers to make healthy choices.”
(We learned after the Sept. Conference that National Milk Producers Federation and the National Dairy Council, funded by the mandatory dairy farmer checkoff, were invited to attend. They were represented, and they brought “student leaders” from GENYOUth. To read NMPF’s statement after the Conference, click here).
Key questions around “what are those healthy choices” to be compassed in tools and identified in FDA labeling went repeatedly unanswered as the discussions focused on approaches and processes, perhaps deeming the unsettled dietary science on fats to be settled science with no need for discussion.
Nutrition Coalition founder, advocate, author and investigative journalist Nina Teicholz has been writing about the Conference for weeks before it began, noting the lack of a pre-conference agenda and the refusal of the Administration to review the science on saturated fats ahead of this ‘landmark’ event.
She points out that the White House delegated Conference planning to the Dean of the Tufts Friedman School of Nutrition Science and Policy at Tufts University Professor Dariush Mozaffarian — developer of the Food Compass, which is a new method for rating and ranking foods in categories to be consumed frequently, modestly, and occasionally.
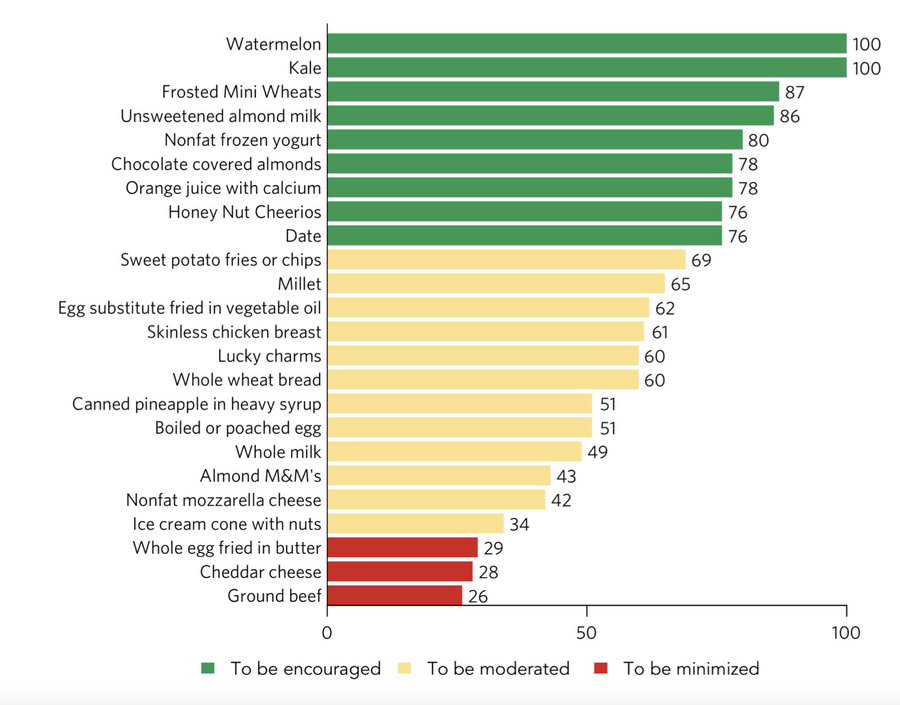
To understand what the Food Compass looks like — sugary cereals rank far ahead of the milk that goes in the bowl with them. And, nearly 70 brand-named cereals from General Mills, Kellogg’s, and Post are ranked twice as high as eggs cooked in butter! Alternative fake milk beverages, such as almond juice, rank ahead of skim milk and far ahead of whole milk. Potato chips (yes, potato chips) are an example of a food that ranks ahead of a simple hard-boiled egg and light-years ahead of whole milk, most cheeses and real beef.
In fact, the only cattle-derived product to get top sector ranking is plain non-fat yogurt. (Surprise: Danone was one of the Food Compass development sponsors). Meanwhile, most cheeses, whole milk, and beef ranked near or at the very bottom of the lowest categories.
Coincidentally, Mozaffarian’s department at Tufts also received a $10 million grant from USDA in November 2021 for a five-year project “to help develop cultivated meat” (aka lab-created meat) through assessment of consumer attitudes and development of K-12 curriculum.
Teicholz laments the lack of consideration by the White House, USDA, HHS and FDA as they ignore many reviews including the most recent state-of-the-art review on saturated fats, whose authors include five former members of the Dietary Guidelines Advisory Committee.
“These are the people who wrote the guidelines saying: ‘We got it wrong,’” writes Teicholz.
Their paper was published in the prestigious Journal of the American College of Cardiologists, whose Editor in Chief named it as one of the top 5 papers of the year. Science like this appears to be off the menu of the White House nutrition playbook.
The entire playbook hinges upon the main tenets of the current Dietary Guidelines for Americans even though the DGAs are being questioned by the scientific community… Even though the DGAs have screened out sound science on dietary animal fats and proteins for at least the past three cycles (15 years)… Even though the rates of American obesity and diet-related illnesses were mostly stable pre-DGA but have risen steadily since the DGA cycles began… And even though these consequences have risen dramatically among children and teens during the past decade since school meals, school milk and a la carte competing foods and beverages were further restricted to the low-fat levels of the DGAs.
What does the White House blame for this poor performance? The playbook cites the Covid pandemic food choices of Americans — stuck at home — for the deteriorated statistics. Unbelievable! These statistics have been deteriorating for decades, especially since 2012.
Looking over the playbook, it closely follows the pattern of FDA’s Multi-year Nutrition Innovation Strategy proceedings that have been quietly underway after public hearings in 2018-19 until the ‘Healthy’ label proposal was announced Sept. 28, 2022.
Appearing in the White House playbook is the proclamation that food and beverage packaging will move toward simpler nutrition guidance under FDA, that an easily recognizable ‘healthy symbol’ will be reserved for front-of-package labeling on those foods the government deems Americans should eat, and a potential ranking system for symbols will be developed for packaging of foods and beverages the federal government deems unhealthy.
This is all coincidentally similar to the Tufts Food Compass, and the substance behind these simplified ‘healthy’ (or not) symbols is a doubling-down on the low-fat DGAs as a primary base metric. Here is a deep dive into the Tufts Food Compass that Mozaffarian, the White House Nutrition Conference Chairman, had a critical role in developing to now be the formation of future food policy. Read the comprehensive analysis here
The National Strategy calls for even more adherence to the flawed DGAs among every sector of the economy beyond government feeding programs, schools, hospitals, and military diets to include foodservice offerings, supermarket layouts, online shopping algorithms, even licensing for all daycare or childcare providers and nutrition certification for these licensed childcare providers – not just those receiving government subsidies for food.
This is so-called “stealth-health” at its best — or rather its worst.
The Biden Administration professes to be concerned about the 1 in 10 households experiencing food insecurity and the rise in diet-related diseases among the leading causes of death and disability in the U.S. The White House cites data showing 19 states have obesity prevalence at 35% or higher with 1 in 10 citizens having diabetes, 1 in 3 with cancer in their lifetime, and nearly 5 in 10 with high blood pressure.
Yet, there is no pause for a comprehensive review of the very dietary guidance, the DGAs, that helped get us here.
The National Strategy reveals how the Administration is assembling executive orders, legislative prompts, calls for action among food organizations, companies, agencies, academia and state and local governments to get everyone on the same page making Davos-style pledges and to conform to the federal playbook.
In the executive summary, the President writes: “Everyone has an important role to play in addressing these challenges: local, State, territory and Tribal governments; Congress; the private sector; civil society; agricultural workers; philanthropists; academics; and of course, the Federal Government.”
(Note Biden’s only reference to farmers or food producers is as “agricultural workers.”)
The playbook’s five pillars talk about improvement, integration, empowerment, support and enhancement. It coins phrases like ‘food as medicine’ and ‘prescriptions for food.’ Reading deeper, we see a launch pad for a new method of nutrition ranking and labeling with the primary factors listed as low-sodium, low-fat and reduced added sugars.
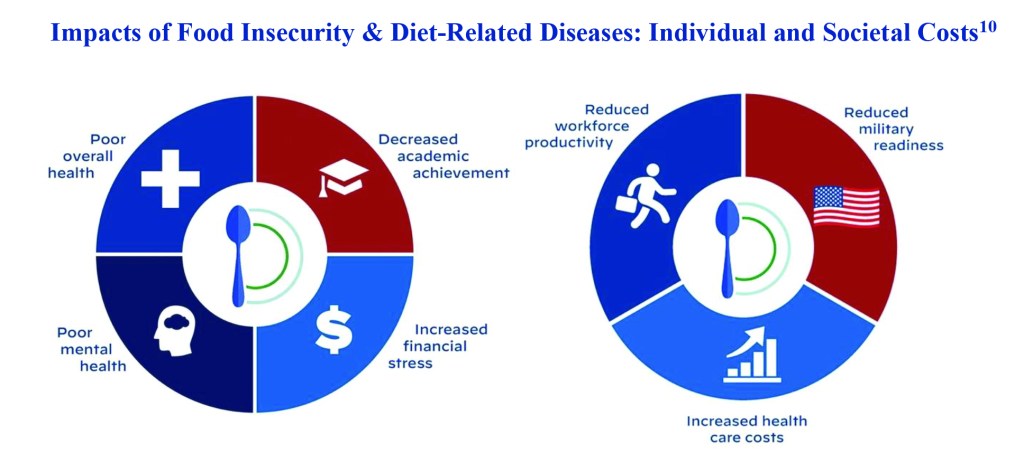
The playbook’s diagrams show us the concerning impact of food insecurity and diet-related diseases in poor overall health, poor mental health, increased financial stress, decreased academic achievement, reduced workforce productivity, increased health care costs and reduced military readiness – but then doubles-down on the solution being more of the same low-fat / high-carb dietary path that got us here.
The White House playbook states that, “The vast majority of Americans do not eat enough vegetables, fruits or whole grains and eat too much saturated fat, sodium and added sugars.” But at the same time, on the saturated fat question, the data show per capita consumption of red meat has declined since the start of the DGAs, and milk consumption has substantially declined.
Americans are being called upon to “unify around a transformational vision,” said Biden.
This vision includes more federal control of diets and nutrition education after failing miserably with the control it already possesses. There is no talk of revisiting the path we are on, just doubling-down on how to get more Americans onto that DGA path, to tell them what to eat, and to put the FDA stamp on ‘approved’ foods and beverages while having the FTC investigate health and nutrition claims that fall outside of the flawed DGAs.
Translation: Let the ‘nutrition bullying’ from the White House bully-pulpit begin. Some of us are ready to rumble.
-30-

 By Sherry Bunting, from Farmshine, August 17, 2018
By Sherry Bunting, from Farmshine, August 17, 2018