UPDATE: The comment period at the Federal Register has been extended an additional 30 days to December 2, 2021
By Sherry Bunting, Farmshine series
WASHINGTON, D.C. — How should ‘cell cultured meats’ be labeled? That’s a loaded question considering how many unknowns surround the commercial production of these lab-grown lookalikes — starting with what are they, really?
USDA Food Safety and Inspection Service (FSIS) announced a 60-day comment period as part of its advance notice of proposed rulemaking in the Federal Register Friday, Sept. 3. The agency seeks “specific types of comments and information that will inform the process of developing labeling regulations for meat and poultry products made using animal cell culture technology.”
Comments are now due by Dec. 2, 2021 and must reference Docket FSIS-2020-0036.
They can be submitted directly here or by going online at the Federal eRulemaking Portal at https://www.regulations.gov and following the on-line instructions; or mail comments to Docket Clerk, U.S. Department of Agriculture, Food Safety and Inspection Service, 1400 Independence Avenue SW, Mailstop 3768, Washington DC 20250-3700.
In a press release, FSIS officials said ‘cell culture meat’ is a terminology the federal agencies use internally, but this is not necessarily the nomenclature to be used in consumer product labeling.
The actual Federal Register notice is lengthy, explaining that the labels for cell culture products fall under FSIS jurisdiction and “will be subject to premarket review under the same process as other special statements or claims. This will ensure that labeling for products developed using cell culture technology are not false or misleading, that labeling requirements are applied consistently as these novel products enter the marketplace, and that the label provides the necessary product information for consumers to make informed purchasing decisions.”
To-date, FSIS has already provided for a “generic approval” of labeling features, statements, and claims based on “demonstrated prevalent industry understanding of the effective application of those features, statements, or claims and consumer understanding of labeling statements.”
However, the document also notes that there is currently “no widespread industry understanding of the labeling requirements for cell cultured meat and poultry products” and that “consumers have not yet had experience reading these types of labels.”
Furthermore, FSIS will have to determine a process for approving additional claims on the labels of these new and combined products.
The docket language suggests that FSIS already considers these proteins analogous as derivatives of the animals from which the original cells are sourced. But are they? Even scientists debate this assumption.
As billionaire-invested startups have joint-ventured with some of the world’s largest food processing companies, much money is being thrown at certain technology hurdles to avoid having to explain the unsavory aspects of the cell culture process to the public — as these lab-grown un-natural proteins inch their way closer to commercial market entry, especially on boneless products like ground beef and chicken tenders and patties.
The label rulemaking step comes two years after the FDA and USDA entered into a joint agreement to each take responsibility for different halves of the ‘cell culture’ process.
The March 2019 agreement came after a summer 2018 public meeting previously reported in Farmshine, for which thousands of comments and two petitions have been logged.
In 2018, the U.S. Cattlemen’s Association (USCA) filed a petition requesting that FSIS limit the definition of ‘‘beef’’ to products derived from cattle born, raised, and harvested in the traditional manner, and thereby prohibit foods comprised of or containing cultured animal cells from being labeled as ‘‘beef.’’ The petition similarly requested the same for the definition of “meat” and other common meat terms on labels.
In 2020, FSIS received a petition from the Harvard Law School Animal Law and Policy Clinic requesting adoption of a labeling approach that “respects First Amendment commercial speech protections” and specifically establishes “a labeling approach that does not require new standards of identity and does not ban the use of common or usual meat or poultry terms.”
This came after over 6000 comments were received on the U.S. Cattlemen’s petition.
In the current rulemaking docket, FSIS states that the comments came from trade associations, consumer advocacy groups, businesses operating in the meat, poultry, and cultured food product markets, and consumers with “most comments opposing the (cattlemen’s) petition overall; however, nearly all generally agreed that cultured meat and beef should be labeled in a manner that indicates how it was produced and differentiates it from slaughtered meat products.”
To some, that kind of interpretation would mean ‘cultured beef without the cow’; to others a better definition would be ‘un-natural beef grown from gene-edited, growth-hormone-promoted laboratory cell cultures.’
Here’s the problem. The lengthy Federal Register docket does very little to explain the real process by which cell cultured un-natural protein is designed and grown before it is harvested, processed and packaged.
The docket includes a description of ‘cell culture’ meat and poultry that fails to specify any of the characteristics, even those that are being questioned by experts in science journals – things that consumers should know and understand via crystal-clear differentiation.
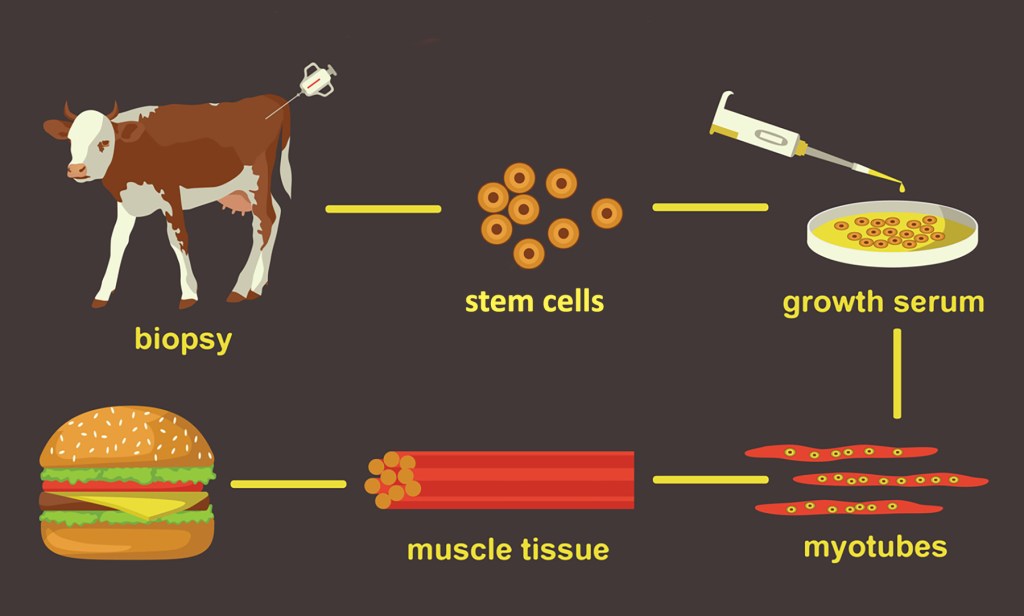
For example, cell culture fake-meat comes from stem cells that are identified and separated from muscle tissue of cattle, pigs, poultry and certain fish. New “continuous cell lines” are being developed from these stem cells using “transformation” processes (gene editing) to make them “immortal.”
In other words, cells normally have a finite end to their growth, but continuous cell lines — under the right controlled environments — are ‘designed’ to keep dividing and growing, continuously, like a malignancy, without an end point.
Also, the ‘growth medium’ for these ‘cell cultures’ contains Fetal Bovine Serum (FBS), growth promoting hormones, and, when needed, antibiotics and fungicides. The controlled environment provides the exchange of oxygen and carbon dioxide akin to animal respiration, and the temperature must be warmed constantly to be the internal temperature of the bovine to keep the cells from dying. At a certain juncture in the process, the growing cells must be ‘fed’ amino acids and carbohydrates.
Reviews of chemical replacements for Fetal Bovine Serum (FBS) are mixed. Some showed the continuation of cell growth was not consistent. Others showed changes happened within the cells when the growth medium included artificial replacements for the FBS. Portions of the veterinary and medical industries also rely on FBS for culturing, and some reports indicate increased importation of FBS, already, for those uses.
Any label claims about nutrition, environmental footprint, possible changes to the actual cells due to the composition of the growth medium, and so forth, are all based on smaller-scale laboratory observation and scale speculation, while consumers have literally zero understanding of the process, and some scientists even question whether the nutrition profiles, taste and texture are similar enough to meet consumer expectations for real meat and poultry.
These are standards of identity issues.
Here’s the other key issue for USDA’s rulemaking on ‘cell cultured meat’ labeling… USDA Food Safety and Inspection Service (FSIS) only regulates the back half of the equation. In March 2019, the agreement between USDA and FDA was to “jointly oversee the production of human food products made using animal cell culture technology and derived from the cells of livestock and poultry to ensure that such products brought to market are safe, unadulterated and truthfully labeled.”
Specifically, this agreement delegates the oversight of cell collection, growth and differentiation to the Food and Drug Administration (FDA). Then, at the stage of “harvest” FDA transfers oversight to USDA’s FSIS, which oversees the cell harvest, processing, packaging and labeling of the products.
According to the FSIS rulemaking notice, the agency believes its current food safety and HACCP systems for real meat and poultry are already “sufficient” to be “immediately applied” to the harvest, processing and packaging of these lab-grown lookalikes and that they are only looking at this final labeling piece. This gives us a clue where the labeling is headed.
Specifically, FSIS seeks comments and information from stakeholders over the next 60 days regarding these key areas of the labeling process:
— Consumer expectations about the labeling of these products, especially in light of the nutritional composition and organoleptic qualities (taste, color, odor, or texture) of the products;
— Names for these products that would be neither false nor misleading;
— Economic data; and
— Any consumer research related to labeling nomenclature for products made using animal cell culture technology.
It will be difficult for true consumer advocacy groups (not meat and poultry industry trade groups who are mostly on board for the mix-and-match) to fully consider their views on the above questions. This is further blurred by the oversimplified FSIS description of the cell culture process that does not include any reference to specific characteristics.
For example, the definition does not mention hormones as inputs, it mentions ‘growth factors’. It doesn’t talk about continuously dividing cell lines, but rather ‘creating food’.
In another section, it doesn’t mention FBS, hormones, antibiotics as inputs but rather simply states: “cells are retrieved and placed in a controlled environment with appropriate nutrients and ‘other factors to support growth’ and cellular multiplication. After the cells have multiplied, ‘additional inputs such as growth factors,’ new surfaces for cell attachment, and additional nutrients are added to the controlled environment to enable the cells to differentiate into various cell types.”
The use of innocent code words belie the specifics.
Of course, states FSIS about the process: “Once produced, the harvested cells can be processed, packaged, and marketed in the same, or similar, manner as slaughtered meat and poultry products.”
Nowhere in this description does it mention the gene editing of the cells to get them to transform for continuous multiplication and growth, nor what evidence exists that consuming such cells is safe. Consumers will want to know what they might be consuming once the world’s largest meat processors begin to use cultured cells as real meat extenders, diluters and substitutes.
Nowhere in this description does it mention the hormones and growth promotants that are the necessary “growth factor inputs” because the cells are growing on their own without the animal’s body, designed by God, to provide the natural hormones for natural growth with natural end points.
Nowhere in this description does the docket mention other clear differences between ‘cell cultured’ un-natural protein vs. real natural meat and poultry. The description suggests they are ‘designer’ derivatives of the real thing, opening the door to claims of being more efficient with less environmental impact. Based on what? A reduction in cattle and other livestock numbers?
Like we’ve seen in dairy with plant-based fakes and lack of standards enforcement by FDA, these ‘novel’ products will get to do the more-than / less-than comparative marketing off the real natural standard while consumers assume all other aspects are equal – when clearly they are not.
Scientific journals such as Frontiers in Nutrition have published scholarly articles pointing out the speculation involved in what this process will look like at commercial scale and what impact it will have on the nutrient characteristics, especially micronutrients like iron and B12, that come from the animal’s interaction with its natural environment. (Even the scaffolds the cells grow on will have methods for stretching cell blobs to simulate movement.)
Some scholarly articles point out that even the environmental claims are suspect because land and water use comparisons for cattle are predominantly what is used in feed production. The lab-grown cell cultures will also have to be “fed”. But they won’t spend part of their ‘lifecycle’ grooming carbon-sequestering grasslands or contributing to planet health in the biogenic carbon cycle.
Furthermore, writes one scientist, the warming required for these cell cultures to grow in bioreactors also create CO2 emissions that are long-lived — potentially adding to the buildup of long-term GHG, whereas the methane emitted from real cattle is short-lived and in fact stable and declining when viewed on a total nutrients per animal basis vs. history. This means, what is seen as a reduction in CO2 equivalents for methane based on the short-term heat-trapping side could be more than lost on the long-term CO2 buildup side, a tough fix down the road.
The problem with climate and environmental label claims is that they are based on speculation about unknowns for un-natural cell culture proteins and are compared to only part of the real story about real natural livestock.
All of these unanswered questions should be part of any USDA FSIS rulemaking process on labeling. These proteins should be labeled as ‘experimental’ and ‘un-natural’ until processes are widely known and understood by scientists, agencies, industry and consumers.
In the Sept. 2 press release, USDA Deputy Under Secretary for Food Safety, Sandra Eskin, states that, “The (proposed rulemaking) is an important step forward in ensuring the appropriate labeling of meat and poultry products made using animal cell culture technology. We want to hear from stakeholders and will consider their comments as we work on a proposed regulation for labeling these products.”
Perhaps what USDA needs to hear from commenters over the next 60 days is that there is not enough public information about how these un-natural proteins are sourced, grown, and gene-edited — or their true nutritional and environmental profiles — to call them beef or meat with a simple qualifying statement few will truly understand.
Proponents of labeling cell culture proteins as meat because the cells are derivative are already whining to FSIS about how new labeling procedures or standards of identity would “stifle innovation.”
Individuals, businesses and organizations should be standing up for the consumer’s right to know what they are consuming and what production processes they are supporting – un-natural cell factories or natural meat raised by farmers and ranchers. There are also consumer health and nutrition questions on the FDA front end that the labeling needs to address accurately on the FSIS back end.
Just because the initial cells come from a cow or a chicken or a pig, doesn’t mean the un-natural ‘culturing’ process and resulting blobs of cells, once consumed, will behave in our bodies like — or contain the same properties as — natural muscle meat from a cow or a chicken or a pig.
Processors will be able to swap a percentage of this for that and barely change their labels if new standards or full descriptions are not used.
Labeling should not give the appearance that this is simply meat without the animal. Some would argue this is Frankenfood. Some would argue this is experimental protein that should have to go through rigorous safety tests on the long-term impacts to health and nutrition. But the climate urgency of the United Nations Food Summit this month is already alluding to fast-tracking these “innovations”, applauding Singapore and China for moving forward most aggressively… to save the planet of course.
Perhaps the question to ask is this: How will labeling clearly differentiate so consumers have a clear choice and farmers and ranchers have a real chance…
The dairy industry is facing this music on its own score with the FDA currently evaluating standards of identity for milk and dairy and looking at the new bovine DNA-altered yeast/fungi/bacteria excrement posing as dairy protein analogs without the cow. Through a process that is in some ways different and in other ways similar to cell culture proteins, the bioengineered yeast excrements are being called “designer proteins from precision fermentation.”
The latest marketing twist is to say the bioengineered yeast are “10 to 20 times more efficient feed converters than cows.” These proteins are already being marketed to global processors of dairy foods as ‘stretchers’ and ‘functional’ ingredients, even as ‘carbon footprint enhancers.’
The economic concern for producers on both counts – meat and dairy – is dilution of their products and captive supply price-control of their ‘markets’.
The concern for consumers is the long-term healthfulness and safety of these ingredients and the increased potential for global food control in the hands of a few, with China already figuring prominently in the protein concentration manufacturing industry, globally.
This labeling discussion is too important to ignore, too important to allow oversimplification. Some in the industry say we must encourage and work beside these new forms of food production to end hunger, control climate change and feed everyone in the future. But the foundation premises of these beliefs are not settled science.
The simple play here, by the tech sector to align and dominate the food industry, is to position these un-natural proteins as helpful analogs grown or cultured or fermented without the animals, that these products are needed to supplement animal-sources and reduce environmental impact of livestock, that climate change urgency requires regulatory fast-tracking, and that simple process-qualifiers on a label will differentiate it while making it palatable to consumers.
Will consumers be led to believe these “innovations” are in all other ways the same as the real thing… when in fact they are not?
-30-
